
Silicone fluid is extremely stable against thermal oxidation. For example, the dimethyl silicone fluid DM-FLUID is virtually unchanged when exposed to temperatures up to 150°C in air. Methylphenyl silicone fluid exhibits even better heat resistance than dimethyl silicone fluid and can be used for long durations in environments reaching 250°C.

Silicone fluid has excellent low-temperature resistance. DM-FLUID remains fluid even at -50°C. Methylphenyl silicone fluid was specially developed for low-temperature applications, so it remains fluid even in environments of -65°C. Since temperature has little effect on viscosity, these products are ideal for applications in cold regions.
Silicone fluid shows little change in viscosity due to changes in temperature. It is thus ideal for use as instrument oil in instruments used in airplanes, automobiles, and trains, and for other applications that make use of silicone fluid's viscous properties. Low-viscosity (10 mm2/s and below) products aside, DM-FLUID offers the advantage of less temperature-dependent viscosity change than common mineral and synthetic oils or other silicone fluids.
The thermal conductivity of dimethyl silicone fluid is lower than that of common organic compounds (nearly the same as that of benzene and toluene) and is roughly 25% that of water. Although thermal conductivity increases with higher viscosities, it becomes nearly constant at viscosities above 100 mm2/s. The thermal conductivity of DM-FLUID is 2.4-3.5x10-4 cal/cm·sec·°C at viscosities below 20 mm2/s, and roughly 3.7-3.8x10-4 cal/cm·sec·°C at viscosities over 50 mm2/s.
The specific heat of silicone fluid varies somewhat depending on viscosity, but is generally about one-third that of water. The values are comparable to common organic compounds that have low specific heat. The specific heat of DM-FLUID is 0.39-0.47 cal/g·°C for viscosities below 20 mm2/s, and approximately 0.36 cal/g·°C at viscosities over 100 mm2/s.
Silicone fluid is nearly chemically inert and is virtually unaffected by 10% alkaline aqueous solutions or 30% acid aqueous solutions at ambient temperatures. However, viscosity increases and gelation can occur if even trace amounts of acids or alkalis are introduced at high temperatures. Silicone fluid is almost unaffected by the presence of aluminum, stainless steel, and most other metals. However, lead, selenium, and tellurium can cause gelation. Precautions must therefore be taken during handling.

Silicone fluid has no adverse effect on metals, nor on most other substances. However, it may reduce the volume and weight of some rubber and plastic compounds due to extraction of the plasticizers when subjected to high temperatures. This tendency is especially strong for low-viscosity fluids. Particular care must be exercised when silicone fluid comes into contact with rubber sealing materials.

The surface tension of silicone fluid is much lower than that of water or common synthetic oils. Silicone fluids spread easily over the surfaces of various substances, and are thus widely used as release agents, defoamers, and ingredients for cosmetics.
| Liquid | Surface tension (mN/m) |
|---|---|
| DM-FLUID | 16 to 21 |
| Mineral oil | 29.7 |
| Water | 72 |

The application of silicone fluid to mold surfaces prevents the adhesion of other materials, thereby enhancing mold releasability. Silicone fluids are widely used as release agents because they have excellent heat resistance and do not contaminate molds or molded materials.

Added in small amounts, silicone fluid has excellent defoaming action. Silicone fluid is primarily used as a defoamer for oil-based foaming fluids.

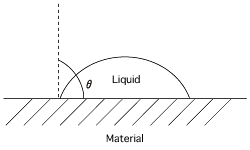
Silicone fluid also has excellent water repellency. The degree of water repellency can be represented by water contact angle, which is over 90° for DM-FLUID. An excellent and durable water-repellent coating can be obtained by applying F-9W-9 or DM-FLUID to glass, ceramic, or fiber using a bake-on process. Silicone fluid can also be used to improve the fluidity of powders and prevent coagulation.
Generally speaking, silicone fluid is physiologically inert and poses almost no risk to people or animals. Low-viscosity products excepted, DM-FLUID is nearly harmless unless ingested in large quantities. It is thus widely used as an ingredient in cosmetics.
The electrical properties of silicone fluid are extremely stable with respect to variations in temperature and frequency. Silicone fluid also withstands dielectric breakdown better than mineral oil. However, as with ordinary insulating oils, moisture absorption affects the electrical insulation properties of silicone fluid, so care must be taken to limit contact with moisture. Steps should be taken to dehydrate silicone fluid before using it as insulating oil in high-voltage transformers.
Silicone fluid has extremely high shear resistance, and it resists shear degradation at high speeds and high loads, meaning it has a long operating life. When synthetic or mineral oils pass through narrow gaps under pressure, shear stress destroys the oil molecules, causing a drop in viscosity. In contrast, with dimethyl silicone fluid of viscosities below 1,000 mm2/s, there is almost no change in viscosity. However, with high-viscosity products there may be an apparent drop in viscosity depending on the shear velocity. Note that this is not due to destruction of the molecules, so the viscosity returns to the initial level when the shear stress is removed. The shear resistance of silicone fluids is over 20 times greater than that of even top-quality petroleum-based oils.
Unlike mineral oil, silicone fluid exhibits extremely high compressibility and does not coagulate when pressurized. Silicone fluid has much higher compressibility than petroleum-based insulating oils or synthetic lubricants, and is thus well-suited for use as damper oil.
Silicone fluid is highly soluble in hydrocarbon solvents such as toluene, xylene, ligroin, and mineral spirits as well as in chlorinated hydrocarbons. However, it is insoluble in ethanol, methanol, and water.
*KF-56 is soluble in ethanol.
Although silicone fluid has many advantageous properties (eg. stable viscosity, high- and low-temperature resistance) that make it ideal for use as a lubricant, its poor boundary lubrication properties on steel-steel interfaces limit its use as a lubricant for such applications. However, silicone fluid does provide good lubrication for steel-bronze, steel-aluminum, steel-zinc, and wood-wood interfaces and for various combinations of plastics.
In terms of radiation resistance, methylphenyl silicone fluid is superior to dimethyl silicone fluid, and the stability of radiation resistance increases proportionately with the amount of phenyl groups present in the molecules. Due to this property and its resistance to high and low temperatures, methylphenyl silicone fluid is widely used in the high-temperature parts of radiological equipment.
| Material | Metal | Silicone fluid DM-FLUID-50cs | ||||
|---|---|---|---|---|---|---|
| Change in appearance |
Weight change rate (%) |
Volume change rate (%) |
Change in appearance |
Viscosity change rate (%) |
Volume resistivity (Ω·cm) |
|
| Before heated storage | - | - | - | - | - | - |
| After heating to 150°C for 1,500 hours, and before metal is inserted |
- | - | - | No change | 0 | ≥1014 |
| Aluminum | No change | 0 | 0 | |||
| Copper | Moderate discoloration | |||||
| Copper (tinned) | Moderate discoloration | |||||
| SUS 27 | No change | |||||
| Iron | Moderate discoloration | |||||
| Iron (cadmium plated) | No change | |||||
| Iron (nickel plated) | ||||||
| Iron (zinc plated) | ||||||
Testing conditions: immersion for 1,500 hours at 150°C in sealed conditions.
| Contact conditions | Change in appearance of solder |
Silicone fluid DM-FLUID-100cs | |
|---|---|---|---|
| Change in appearance |
Viscosity change rate (%) |
||
| Contact at room temp. | No change | No change | 0 |
| Contact at 70°C | |||
| Contact at 100°C | |||
| Contact at 150°C | |||
Testing conditions: immersion for 500 hours at room temperature, 70°C, 100°C, and 150°C.
| Material | Change in plastic | Change in silicone fluid | |
|---|---|---|---|
| Weight change rate (%) | Volume change rate (%) | Appearance | |
| Polyethylene | -0.02 | -0.09 | No change |
| ABS resin | -014 | -0.16 | |
| Teflon | +0.03 | +0.15 | |
| Hard PVC | 0 | +0.05 | |
| Polystyrene | -0.04 | 0 | |
| Phenol resin | +0.30 | +0.37 | |
| Methacrylic resin | -0.02 | +0.08 | |
| Polycarbonate | +0.03 | 0 | |
| Acetal resin | +0.02 | +0.08 | |
| Nylon | 0 | -0.01 | |
| Cellulose triacetate | +0.01 | +0.05 | |
Testing conditions: immersion for 500 hours at 70°C.
| Material | Contact conditions | Volume chage rete (%) |
|---|---|---|
| Nitrile rubber 1 | 105°C / 250 h | -6.7 |
| Nitrile rubber 2 | -8.5 | |
| Nitrile rubber 3 | 150°C / 200 h | -6.0 |
| Butyl rubber | 105°C / 250 h | -8.3 |
| Styrene butadiene rebber | -5.9 | |
| Chloroprene rebber | -12 | |
| Neoprene rebber | -12 | |
| Ethylene propylene diene polymer | 150°C / 200 h | -12 |
| Acrylic rebber | 150°C / 250 h | -4.3 |
| Fluoro-rebber (Viton®) | +0.8 | |
| Silicone rebber KE-870-U | 150°C / 250 h | +37 |
| Silicone rebber KE-765-U | +41 | |
| Silicone rebber KE-951-U | +50 | |
| Silicone rebber KE-550-U | +51 | |
| FluoroSilicone rebber FE-271-U | +0.5 |
Silicone fluid has major effects on silicone rubber,
with significant swelling of the rubber.
Lower viscosity fluids have greater effects.
In contrast, there is almost no swelling of fluorosilicone rubber.
| Substance | Contact angle (°) |
|---|---|
| Paraffin | 108-116 |
| Carnauba wax | 107-125.3 |
| DM-FLUID | 90-110 |
| Naphthalene | 62 |
| Nylon | 70 |
| Polyethylene | 94 |
| Polyvinyl chloride | 87 |
| Polystyrene | 91 |
| Polytetrafluoroethylene | 108 |
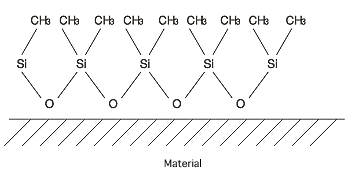
When DM-FLUID is applied to a surface using a bake-on method,
the hydrophobic methyl groups (CH3-, shown below)
face outward, a state which results in water repellency.
Testing method
Silicone fluid was applied on a patch to the inside of a human subjects' upper arm and the reaction observed with a microscope after 24 hours.
Test results
| Grade | Determination |
|---|---|
| DM-FLUIDL-5cs | Quasi-negative |
| DM-FLUIDL-10cs | Negative |
| DM-FLUIDL-100cs | Negative |
Determination standards
| Microscope determination |
Naked eye determination |
|||||
|---|---|---|---|---|---|---|
| Irritation ranking | B irritation (Sample irritation index) - (Control irritation index) |
C irritation | D irritation | |||
| Determination standards | 0 | 1-2 | 3 | ≥4 | One or more instances |
Determination and assessment |
| Determination and assessment |
Negative | Quasi- positive |
Quasi- positive |
Positive | Positive | Positive |
Number of test subjects: 20 (Japanese Society for Cutaneous Health)
Test conditions
| Animal | Japanese white rabbit |
|---|---|
| Sample | DM-FLUID-5cs |
Test results
Absolutely no effect on the cornea or iris.
There is slight inflammation of the conjunctiva,
but to a far lower degree than that caused by typical detergents.
Test conditions
| Animal | rat |
|---|---|
| Sample | DM-FLUID-5cs |
Test results
LD50 is over 5,000 mg/kg for both males and females.
*Acute toxicity test
Generally speaking, this test determines the amount of a substance that constitutes
a lethal dose when administered at one time to a test animal.
It is usually expressed as "LD50" (50% Lethal Dose).
Please refer to the following table of degree of toxicity.
Classification based on strength of toxicity
| Degree of toxicity* | Oral LD50 (rat) (unit: mg/kg) |
|---|---|
| Very toxic | ≤25 |
| Toxic | 25-200 |
| Harmful | 200-2000 |
The arc resistance of silicone fluids is about the same as that of ordinary mineral oil-based insulating oils. Table shows the results of tests conducted to determine the amount of gases emitted and amount of deposits caused by arcing.
| Material | Gas quantity emitted per 1,000 arcs |
Composition of non-oil gas discharges (Vol. %) | Gas deposits (carbon, other) per 1,000 arcs |
|||||||
|---|---|---|---|---|---|---|---|---|---|---|
| H2 | CO | CO2 | CH4 | C2H4 | C2H2 | Except C2H2 CnH2n |
N2+O2 | |||
| Silicone fluid | 9.9 | 50.6 | 2.6 | 0.5 | 5.8 | 0.5 | 0 | 0.5 | 39.5 | 26 |
| Common insulating oil | 6.2 | 48.8 | 1.3 | 0 | 4.5 | 0.7 | 3.3 | 0.7 | 40.7 | 13 |
| Liquid paraffin | 7.0 | 49.4 | 2.4 | 0.4 | 2.4 | 1.2 | 2.4 | 0.6 | 41.2 | 14 |
DC intermittent arc method Applied voltage:
100 V Tungsten electrode (From Degradation of Insulating Oils Due to Arcing, a technical report by the Insulating Oil Division)
| DM-FLUID -50cs |
DM-FLUID -1,000cs |
F-6W-9 | F-9W-9 | KF-965 | F-5W-0 | KF-54 | KF-56 | |
|---|---|---|---|---|---|---|---|---|
| DM-FLUID-50cs |  |
 |
 |
 |
 |
 |
 |
 |
| DM-FLUID-1,000cs |  |
 |
 |
 |
 |
- |  |
 |
| F-6W-9 |  |
 |
 |
 |
 |
 |
 |
 |
| F-9W-9 |  |
 |
 |
 |
 |
 |
 |
 |
| KF-965 |  |
 |
 |
 |
 |
 |
 |
 |
| F-5W-0 |  |
 |
 |
 |
 |
 |
 |
 |
| KF-54 |  |
 |
 |
 |
 |
 |
 |
 |
| KF-56 |  |
 |
 |
 |
 |
 |
 |
 |
 : Dissolves transparently at room temperature
: Dissolves transparently at room temperature
 : Clouds or separates at room temperature
: Clouds or separates at room temperature
| Solvent | Results |
|---|---|
| Toluene |  |
| Xylene | |
| Solvent naphtha | |
| Industrial gasoline | |
| Mineral spirits | |
| Kerosene | |
| Cyclohexane | |
| n-hexane | |
| n-heptane | |
| Diisopropyl ether | |
| Hexyl ether | |
| Ethyl acetate | |
| Butyl acetate | |
| Isopropyl laurate | |
| Isopropyl palmitate | |
| Isopropyl myristate | |
| Methyl ethyl ketone | |
| Methyl isobutyl ketone | |
| Lauryl alcohol | |
| Acetone |  |
| Butanol | |
| 2-ethylhexanol | |
| Amyl acetate | |
| Glacial acetic acid | |
| Naphthene-based lubricating oils | |
| Methanol |  |
| Ethanol | |
| Ethylene glycol | |
| Glycerin | |
| Diethylene glycol stearate | |
| Propylene glycol | |
| Liquid paraffin | |
| Paraffin wax | |
| Petrolatums | |
| Lubricating oils | |
| Fatty acids (other than glacial acetic acid) |
|
| Animal and vegetable oils | |
| Methyl phthalate | |
| Water |
 : Dissolves
: Dissolves
 : Partially dissolves
: Partially dissolves
 : Does not dissolve (results at room temperature)
: Does not dissolve (results at room temperature)